Indications
Enteral feeding should be considered in all patients in whom oral intake is significantly reduced or absent for at least 5-7 days[5] and should be given early consideration in post-surgical patients.
A poorer baseline nutritional status warrants earlier commencement of enteral feeding as early as within 1-2 days post-surgery.
Common indications are listed as follows[6]:-
- Unconscious patients (ventilated/post neurological trauma)
- Swallowing disorders
- Partial intestinal failure
- Pancreatitis
- Increased nutritional requirements (e.g.
cystic fibrosis).
- Psychological disorders (e.g.
Anorexia Nervosa)
Contraindications
Relative contraindications to enteral feeding include intestinal obstruction,
ileus and small bowel fistulae7.
Recent lower gastrointestinal anastomoses are not a contraindication to enteral feeding though a more cautious approach is advised in gastric,
biliary and pancreatic anastomoses[6].
Pre-procedure preparation
1. Diagnostic evaluation – This should be commenced with a review of the clinical indications as described above and an examination of the most recent cross-sectional imaging particularly in cases of luminal stricturing and variant anatomy.
Endoscopic examination reports can prove invaluable in the former and failure to pass a stricture under direct visualisation should prompt earlier consideration of alternative feeding strategies or recommendation to proceed only under direct visualisation to ensure safety.
Operative notes should be routinely consulted in post-surgical patients for details of any anastomoses and alterations in the normal course and position of the alimentary canal (Fig.
2,3).
A brief assessment of the patient’s clinical condition and physiological observations (heart rate,
respiratory rate,
blood pressure and oxygen saturations) will provide a baseline to allow continuous monitoring of the patient in this often critically unwell patient group.

Fig. 2: Nasojejunal tube placed satisfactorily in a patient with previous gastrectomy and primary oesophago-jejunal anastomosis. Subsequent anastamotic stricture traversed by use of the above procedure.
References: NHS Grampian

Fig. 3: Nasojejunal tube placed satisfactorily in a patient with previous gastrectomy and primary oesophago-jejunal anastomosis.
References: NHS Grampian
2. Informed consent and patient preparation – Verbal consent is to be obtained with a discussion about the possible major complications including but not limited to cardiac arrhythmias,
tracheo-bronchial trauma and luminal perforation (see section below).
Minor complications such as tube dislodgement,
procedure failure and repeat attempts should also be highlighted and at a minimum the patient should be made aware of the use of ionising radiation during the procedure.
A brief overview of the procedure should be given to the patient with regards to the expected discomfort and need to maintain specific positions during the procedure.
3. Medication – While there is little role for routine pre-procedural medication ensuring maximal patient comfort prior to commencement is advised with consideration given to parenteral anti-emesis and analgesia in those with intractable nausea and pain which may reduce the patient’s tolerance of the procedure.
Topical Lidocaine administered via a nasal spray at the outset of the procedure along with the use of instillagel to anaesthetise the mucous membranes of the nasal cavity will ease initial passage of equipment.
4. Equipment – A list of required equipment with examples as used at our institution is listed below and in the annotated (Fig.
4).
- a.
Radiopaque Nasojejunal/Nasogastric tube (e.g.
Enfit® Standard Nasojejunal feed/aspiration tube 7/9fr x 130cm or Freka® Easy In Double Lumen Nasojejunal Feeding Tube)
- b.Topical Lidocaine(including nasal adaptor) and Instillagel
- c.
Single lumen nasal cannula for delivery of oxygen into the contralateral naris
- d.
Yaunker suction catheter
- e.
100cm 4/5 Fr Angiographic Vertebral Catheter (e.g.
Radifocus® Glidecath® - Hydrophilic Angiographic Catheter)
- f.
260cm 0.035" angle tipped standard guidewire (e.g.
Radifocus® Guidewire M Standard type)
- g.
260cm 0.035" angle tipped exchange length stiff wire (e.g.
Radifocus® Guidewire M Stiff type)
- h.
Low osmolar contrast medium (eg.
Omnipaque™ 240) safe for oral use diluted 1:1 with 0.9% Saline
- i.
0.9% saline ampoules,
syringes,
gauze and gallipots
- j.
Secure dressing and nasal Bridle (if applicable)
- k.
Cup of water with a straw.

Fig. 4: Prepared equipment tray with annotations.
References: NHS Grampian
Procedural steps (Post-pyloric tube placement)
- Review relevant imaging and previous investigations (e.g.
endoscopy reports etc.).
- Ensure availability of trained nursing staff and at least an hour allotted for the procedure in the fluoroscopy suite.
- Explain the procedure to the patient and gain verbal consent.
Assess the patient’s baseline clinical condition.
Those requiring oxygen at the outset should have continuous pulse oximetry due to the risk of rapid desaturation when gagging or with inadvertent passage of catheters or tubes into the trachea.
Oxygen should be delivered to match requirements continuously via the single pronged nasal cannula.
- Gloves should be worn at all times to ensure a clean field.
- Prepare all equipment taking care to instil saline in the standard and stiff hydrophilic guidewire cases and within the vertebral catheter to allow ease of guidewire passage.
- Begin with the patient seated upright on the fluoroscopy table if tolerated or supported with pillows in a supine position.
- Estimate required enteral tube length against the patient to the approximate position of the duodenum if using an adjustable tube (e.g.
Freka) and cut down to size.
This should be from the nose to the duodenojejunal flexure along the approximate route of the alimentary canal via the earlobe[6].
- Assess the patency of both nares choosing the clearer side and applying topical lidocaine to the nasopharynx with a nasal adaptor.
A small amount of instillagel can also reduce discomfort.
- Insert the vertebral catheter angled caudally and posteriorly with the patient’s head tilted slightly upwards (Fig 5).
The patient should be instructed to begin swallowing when they feel catheter reach the posterior wall of the nasopharynx which can be aided with the patient sipping water.
Patients invariably gag at this point and care must be taken to ensure the catheter is not coiling within the oropharynx or progressing into the trachea.
Excessive sputum produced from pharyngeal irritation should be removed intermittently by suction.
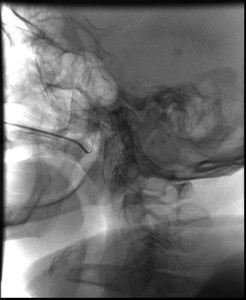
Fig. 5: Vertebral catheter with guidewire in situ entering the pharynx at the start of the procedure.
References: NHS Grampian
- Once the catheter is deemed to have passed beyond the hypopharynx the patient is moved to a recumbent position and the catheter is advanced to the stomach.
- Once within the stomach,
under fluoroscopic control the catheter should be guided along the greater curvature to reach the pylorus (Fig.
6).
The patient can be mobilised partially onto their right side for gravitational assistance.

Fig. 6: NJ tube in situ demonstrating classical anatomy with tube passage against the greater curvature of the stomach towards the pylorus. PEG tube in situ.
References: NHS Grampian
- Fluoroscopy is coned to the site of the distal catheter tip and using pulsed fluoroscopy at rates as low as 1 pulse per second(PPS) can markedly reduce radiation exposure.
Magnification can reduce the dose further and aid accurate direction of the catheter which is steered by applying clockwise/anticlockwise torque to the luer-lock port at the proximal end of the catheter.
- The patient is returned to a supine position once at the antrum and the catheter is manipulated past the pylorus.
As this is the most frequent hurdle (Fig.
7) the 260cm 0.035’’ standard angle tipped guide wire can be used to direct the catheter following instillation of a small amount of diluted contrast to delineate the anatomy.
Longitudinal Rugae and transversal Valvulae Conniventes will differentiate the stomach and small bowel respectively.
The patient can also be turned to their left to straighten the pylorus or a gloved hand can be used to apply cephalad pressure upon the greater curvature[1].
Entry of the catheter into the duodenum is usually confirmed by posterior and cephalad passage followed by rapid caudal descent into the second part of the duodenum.
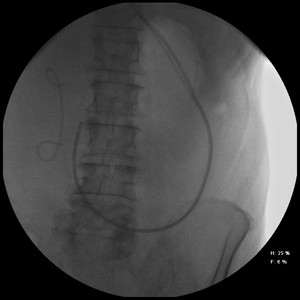
Fig. 7: Failure to navigate the pylorus causing the procedure to be abandoned at this point. Common bile duct stent in situ.
References: NHS Grampian
- Passage of the catheter through the second,
third and fourth parts of the duodenum is usually straight-forward in the absence of a stricture (Fig.
8).
In this event the guidewire and catheter can be used to steer past the obstruction.
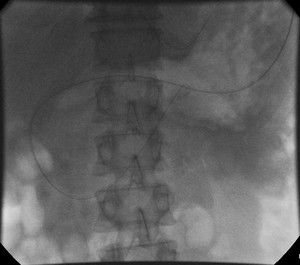
Fig. 8: Catheter over wire in situ till the level of the DJ flexure. Passage of the catheter through the second, third and fourth parts of the duodenum is usually straight-forward in the absence of a stricture
References: NHS Grampian
- A standard vertebral catheter at 100cm is generally sufficient to reach the third/fourth part of the duodenum.
At this point the standard guidewire can be switched for a 260cm 0.035’’ stiff exchange length wire which can be advanced till the duodeno-jejunal flexure which is marked by anterior and caudal passage with deflection to the right or left[1] (Fig.
9)

Fig. 9: Catheter exchanged for NJ tube.
References: NHS Grampian
- The vertebral catheter is now removed leaving the stiff exchange length wire in place with intermittent screening to ensure the tip of the wire is not dislodged during catheter retrieval (Fig 9).

Fig. 10: (left to right) – Catheter exchanged for stiff guidewire to maintain position with NJ tube finally passed over stiff guidewire.
References: NHS Grampian
- A small amount of instillagel is placed and the nasojejunal tube is passed over the stiff wire with its tip beyond the ligament of trietz during which intermittent screening ensures safe tube passage (Fig 11 & 12).
The guide wire can now be removed,
and a small amount of contrast can be instilled to ensure free flow beyond the tube tip (Fig.
13).

Fig. 11: The nasojejunal tube is passed over the stiff wire with its tip beyond the ligament of trietz during which intermittent screening ensures safe tube passage. This image demonstrates coiling of tube in upper oesophagus during placement over stiff guidewire.
References: NHS Grampian

Fig. 12: Satisfactory Catheter placement at DJ flexure with subsequent suboptimal positioning of the NJ tube at the end of the procedure.
References: NHS Grampian

Fig. 13: Contrast instillation to ensure passage into the jejunum
References: NHS Grampian
- A fluoroscopic screen grab (Fig.
14) at this point is sufficient for documentation purposes and exposures are not required.
If the gastric suction portion of the tube is separate (e.g.
Freka®) this is now passed over the enteral lumen and secured to the latter.
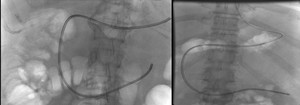
Fig. 14: Satisfactory NJ tube placement in two separate cases.
References: NHS Grampian
- All tube ports should be flushed straight away to remove any retained contrast which may cause blockage.
- Note should be made of length of tube within the patient and included in final documentation and reports.
- Skin on the ipsilateral cheek should be cleaned and an appropriate tube specific dressing should be applied to ensure the tube is held in place.
A nasal bridle can be applied with the patient’s consent if clinically appropriate.
- N.B.
Placement of a nasogastric tube proceeds in a similar initial manner to the above however tube placement can be carried out directly without the use of vertebral catheters or guidewires.
Fluoroscopy confirms tube placement in a safe position within a minimum of 10cm of tube beyond the gastro-oesophageal junction.
The described catheter-guidewire technique is indicated with post-surgical anastomosis and luminal strictures.
Pitfalls and tips
- Difficulty traversing the hypopharynx due to a strong gag reflex can be alleviated by use of fluoroscopy to guide the catheter along the posterior pharyngeal wall and beyond the larynx (Fig.
15).
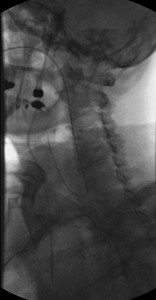
Fig. 15: Catheter coiling within the hypopharynx. Difficulty traversing the hypopharynx due to a strong gag reflex can be alleviated by use of fluoroscopy to guide the catheter along the posterior pharyngeal wall and beyond the larynx
References: NHS Grampian
- A standard 100cm vertebral catheter is often insufficient to get beyond the pylorus due to gross gastric distention commonly seen in this patient population.
This can be alleviated by a period of aspiration via an Anderson’s drainage tube placed in the stomach for several hours preceding the procedure.
Alternatively,
a longer,
non-standard vertebral catheter up to 120cm in length can be used though this may entail higher costs.
- If the stomach is adequately emptied,
gastric air insufflation can propagate peristaltic waves which may aid passage of the vertebral catheter tip across the pylorus.
- Feeding tubes with closed tips and side holes should have the tip cut prior to placement to allow guidewire placement.
This will also allow stiff guidewire placement into the feeding tube in cases of partial tube displacement or proximal migration (Fig 16).
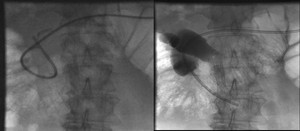
Fig. 16: (Left to right). Images demonstrate a finding of tube displacement with tip lying in the first part of the duodenum. A stiff guidewire was used to straighten the tube and return the tip into the third part of the duodenum. This was made possible by ensuring the distal tube tip was cut prior to initial placement therefore allowing guidewire passage and manipulation.
References: NHS Grampian
Complications
- Tracheobronchial penetration and luminal perforation during the tube placement step represent a small but significant risk[5] as is an extremely rare but documented risk of cardiac arrhythmias (3 in 883 patients in one series)[7].
- Gastro-oesophageal reflux and aspiration of gastric contents may occur with the patient supine due to compromise of the lower oesophageal sphincter with large bore feeding tubes.
These risks may be reduced by slight elevation of the head and the use of proton pump inhibitors during feeding[6].
- Tube blockage – Usually due to infrequent flushing between feeds or the use of non-standard or completely liquid feeds.
High pressure flushing with saline is usually sufficient to relieve this,
failing which an alkaline solution of pancreatic enzymes may be tried[6].
- Tracheo-oesophageal fistula formation – Rare but significant complication may develop when enteral feeding tubes are in situ concurrently with endotracheal airways due to pressure necrosis[5,6].
















